Marble Hcl Reaction

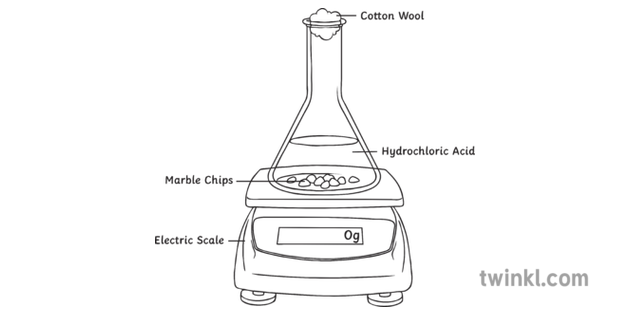
Hydrochloric acid to react with the marble chips independent variable marble chips to react with the acid dependent variable stopwatch to accurately time the experiment spatula to handle the marble chips measuring cylinder to precisely measure out different concentrations of hydryochloric acid electric balance to measure the mass g of the marble chips bung.
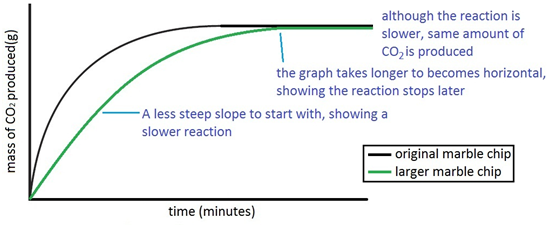
Marble hcl reaction. Marble chips are mostly made up of calcium carbonate which is a alkaline compound. Click each image to see positive and negative results of the acid test. Therefore resulting in a quicker reaction. This is because there will be more hydrochloric acid particles to collide with the marble chip particles.
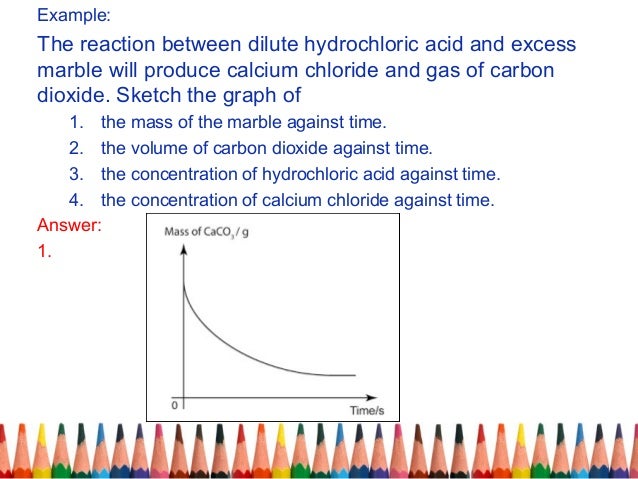
In fact if you take baking soda which contains hco3 and add vinegar which donates h to the solution this is precisely what happens. Calcium carbonate is dissolved by hydrochloric acid thereby forming gaseous carbon dioxide. Marble is crystalized caco3. Marble chips and hydrochloric acid planning aim to find if changing the concentration of an acid will increase or decrease the rate of the reaction when marble is dissolved in hydrochloric acid.
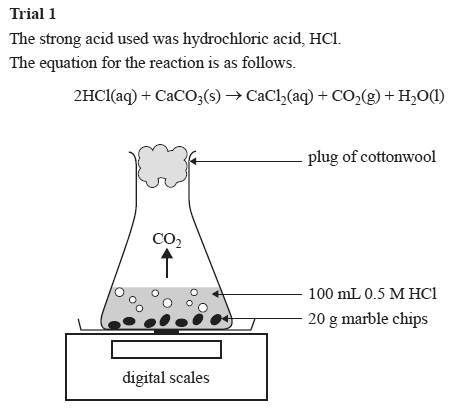
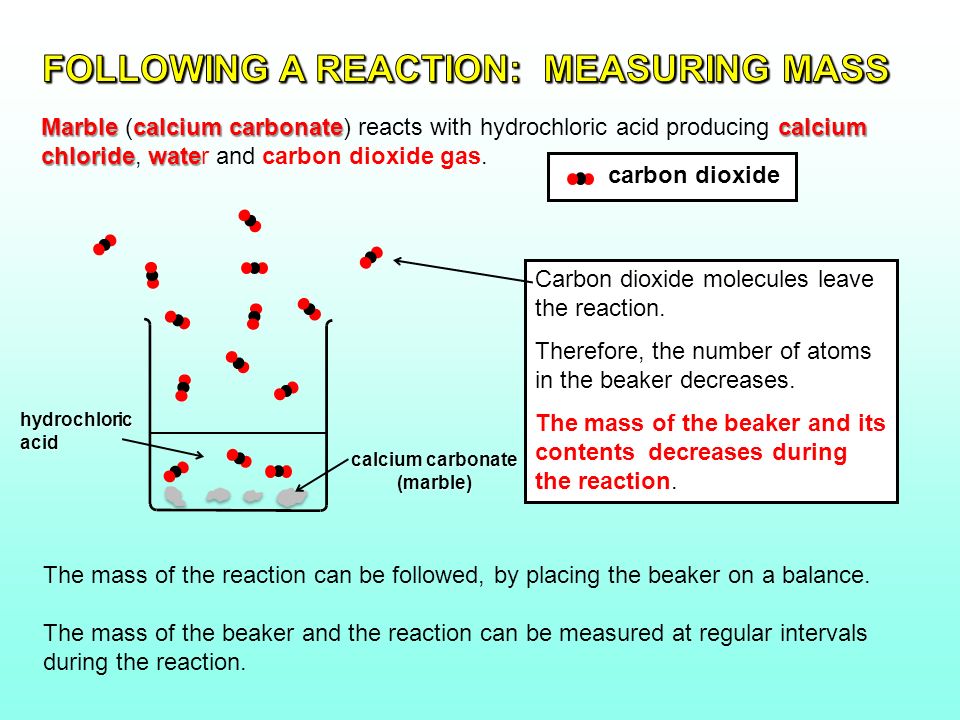
The higher the concentration of hydrochloric acid in the beaker the faster the reaction will take place. The combined reactants have a higher chemical potential than the combined products i e. The overall reaction is 2hcl caco3 cacl2 co2 h2o. Detailed description pdf video mpg4.
Hydrochloric acid or hcl is a strong acid that donates h to water. Pieces of marble are thrown into hydro chloric acid. The increased h increases the rate of the reverse reactions forming carbon dioxide and water. This process is based on random particle movement.
Marble is calcium carbonate and thus behaves in the same way. Marble reaction with hydrochloric acid drop a small amount of dilute hydrochloric acid on an area of your sample that has been scratched by a nail. With the equation caco3 2hcl cacl2 h2o co2 hypotheses a reaction occurs when particles collide. The reaction takes place spontaneously.
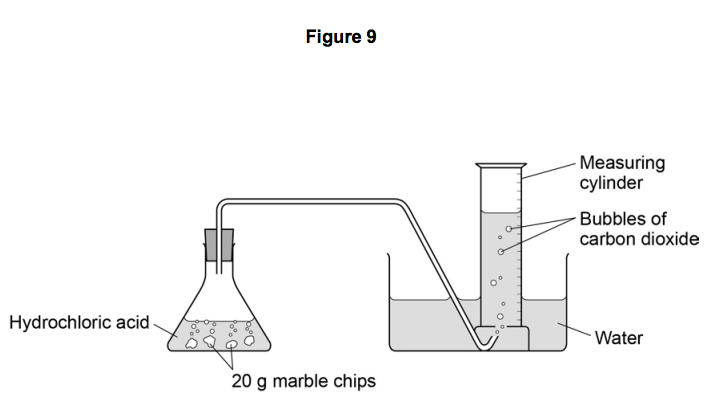
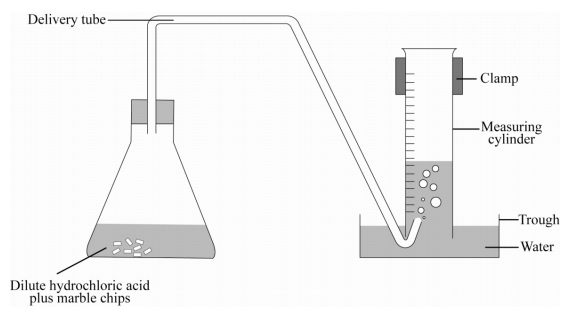
Calcium carbonate and hcl. Cacl2 aq h2o l co2 g in this experiment i am going to see if temperature affects the reaction rate between marble chips and hydrochloric acid by timing the release of carbon dioxide in the reaction. Caco3 s 2hcl aq 61614.