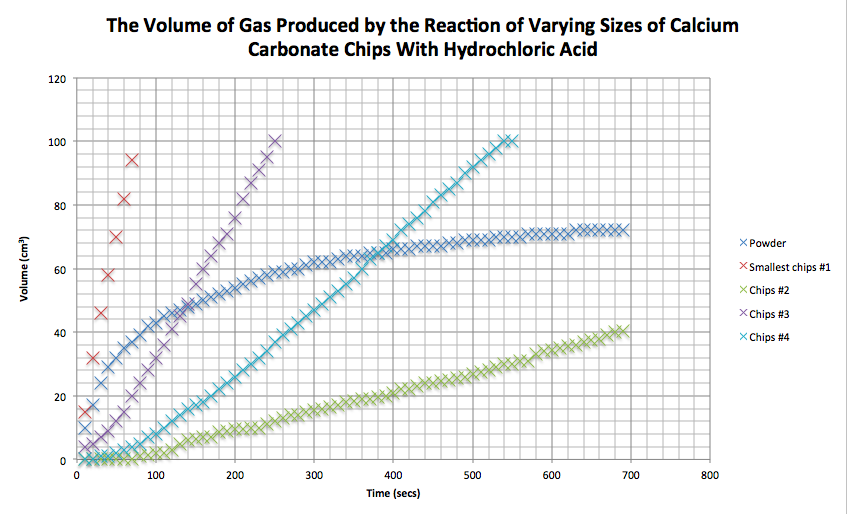
Marble Chips And Hydrochloric Acid Graph

Place 40cm 3 of hydrochloric acid in an conical flask.
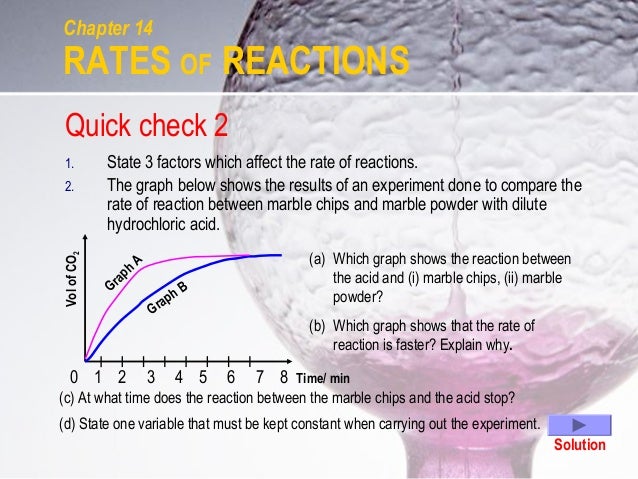
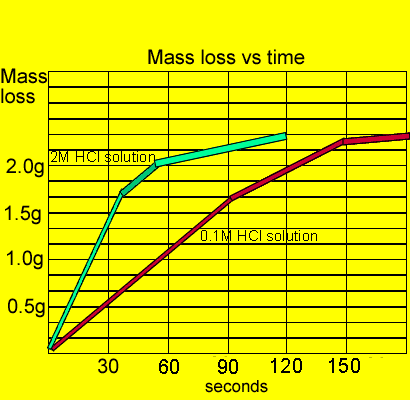
Marble chips and hydrochloric acid graph. 0 5 g of magnesium ribbon and 0 5 g of magnesium powder are reacted with identical samples of hydrochloric acid. Task my task is to measure the rate of reaction between marble chips caco 3 and hydrochloric acid 2 hcl. This is because as time goes on the volume of the gas evolved does not change. Plugged in scientific scales and weighed out 1g of marble chips for each test tube.
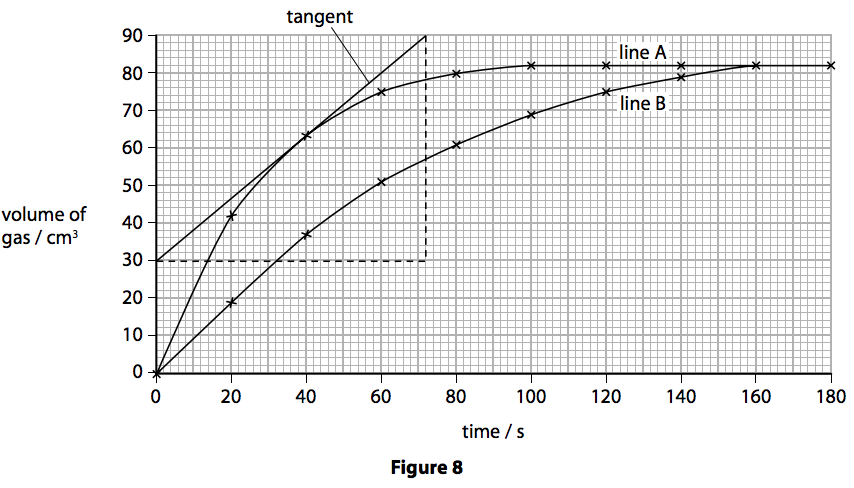
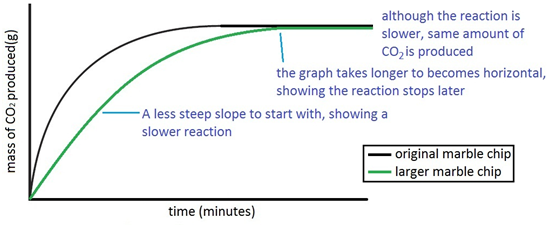
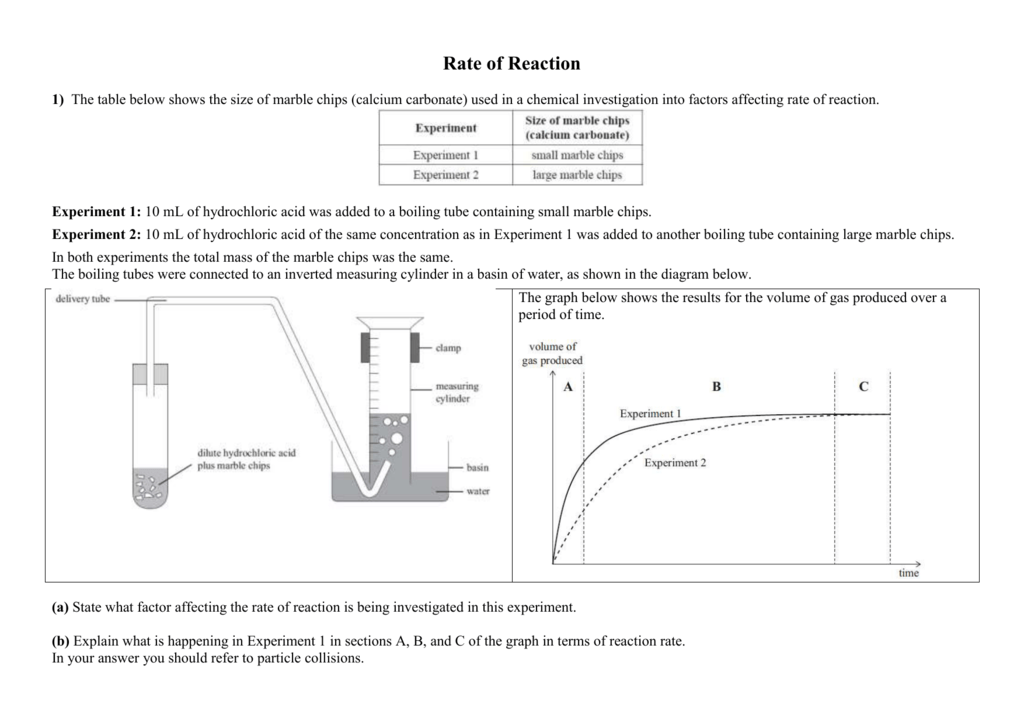
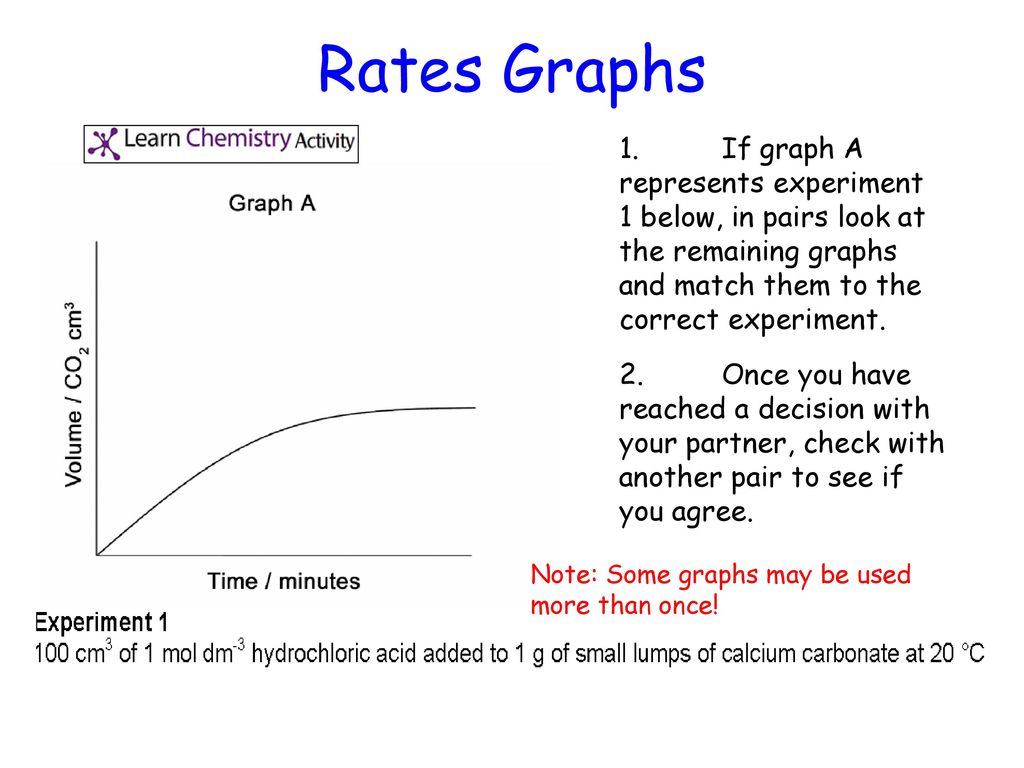
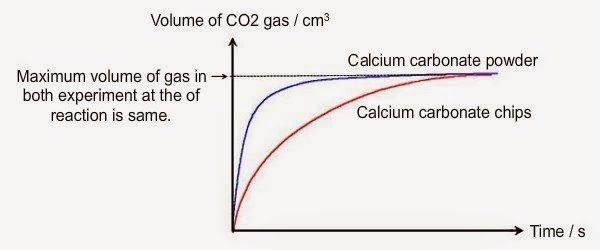
The curve on the graph goes flat when the reaction is complete. The rate of this reaction can be changed by changing the size of the marble chips. Which of these statements explains what would. In the investigation i am going to find out how the surface area affects the rate of reaction by measuring the amount of gas produced and weight loss in a reaction between small large pieces of marble chips calcium carbonate and hydrochloric acid per minute.
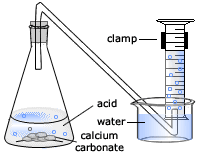
C reaction of marble chips and acid. A conical flask contains the marble chips hydrochloric acid and the water that will make the reaction. Measured 5ml of hydrochloric acid in the 10ml measuring cylinders and placed into each beaker separately. Diagram plan the equipment i will be using for this experiment will be as follows.
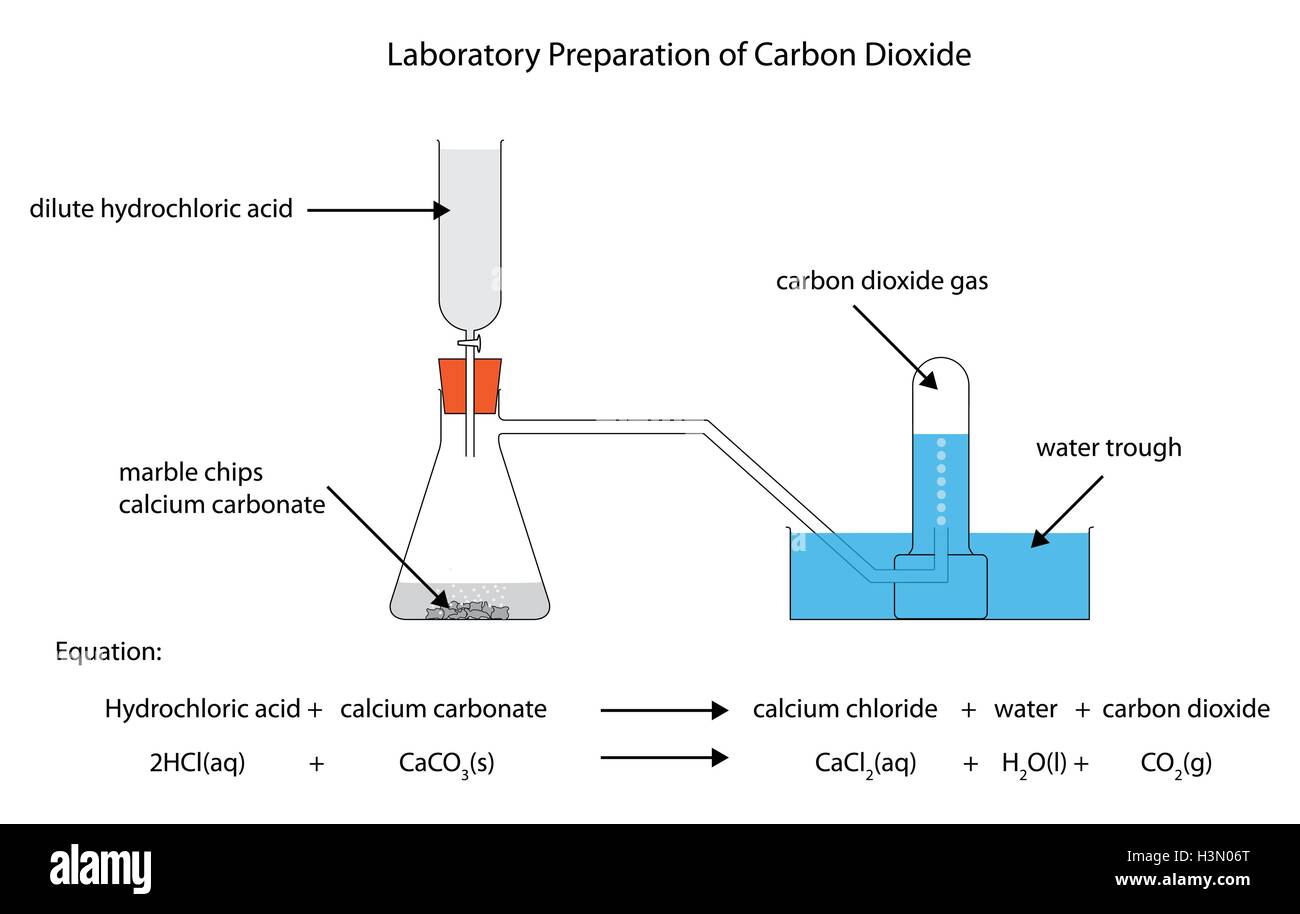
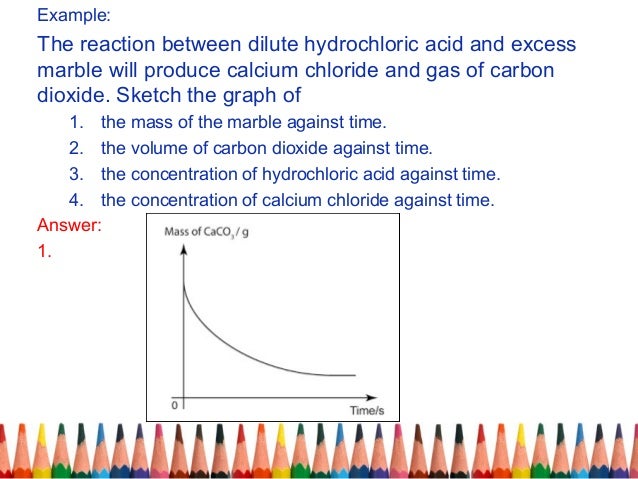
An investigation into how changing one variable influences the rate of reaction between marble chips and dilute hydrochloric acid planning section when dilute hydrochloric acid reacts with marble chips the following reactions occurs. To investigate the effect of concentration on the reaction between marble chips and hydrochloric acid materials. A chemistry investigation to look at the rates of reaction between marble chips and hydrochloric acid. Marble chips react with dilute hydrochloric acid to produce carbon dioxide gas.
Marble chips placed onto pieces of paper. A tube to connect the conical flask to the measuring cylinder. Measured out 1ml of water in a 10ml measuring cylinder and placed into the test tube labelled 2. A stand to hold up the measuring cylinder.
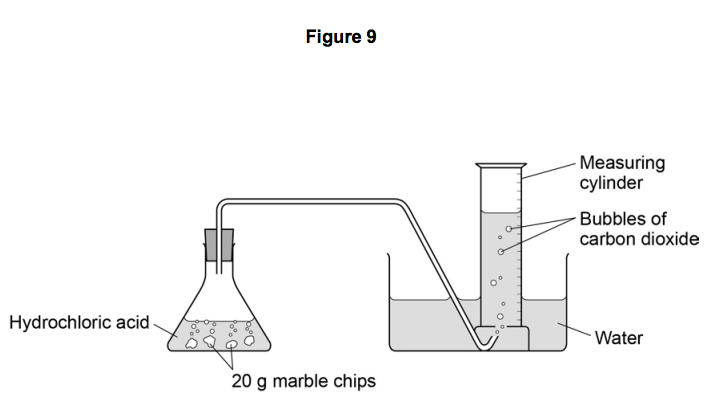
Investigating the rate of reaction between marble chips calcium carbonate and hydrochloric acid aim. Measuring the rate of loss of a gaseous product. Conical flask delivery tube bung measuring cylinder x 2 water trough water stopwatch marble. Caco3 2hcl h2o co2 this is the reaction we will be investigating.
In the reaction between calcium carbonate marble chips and hydrochloric acid we can use the apparatus below to find the rate of reaction.













.jpg)